Overview
remedē® is an FDA-approved, implantable, non-mask therapy for moderate to severe central sleep apnea (CSA) in adult patients. It is sometimes referred to as “phrenic nerve stimulation” because of the way in which the therapy treats central sleep apnea. remedē stimulates a nerve in the chest (phrenic nerve) that sends signals to the breathing muscle (diaphragm) to control breathing. In some ways, it is like a pacemaker for your breathing – it helps your body to maintain a consistent breathing pattern while asleep. This is very different from mask based therapies that work by pushing air in through your mouth or nose to open the airway. Because remedē is implantable and activates automatically, it does not require wearing a mask. Only your doctor can determine if phrenic nerve stimulation is right for you.1
How remedē works
In a healthy patient without CSA, the brain signals the breathing muscle (diaphragm) to breathe in and out at regular intervals. The signal to breathe travels from the brain stem, down the phrenic nerve, and then to the diaphragm, causing a contraction. This contraction of the diaphragm pulls air into the lungs. The problem for patients with central sleep apnea is that the brain fails to send regular signals to the diaphragm.2
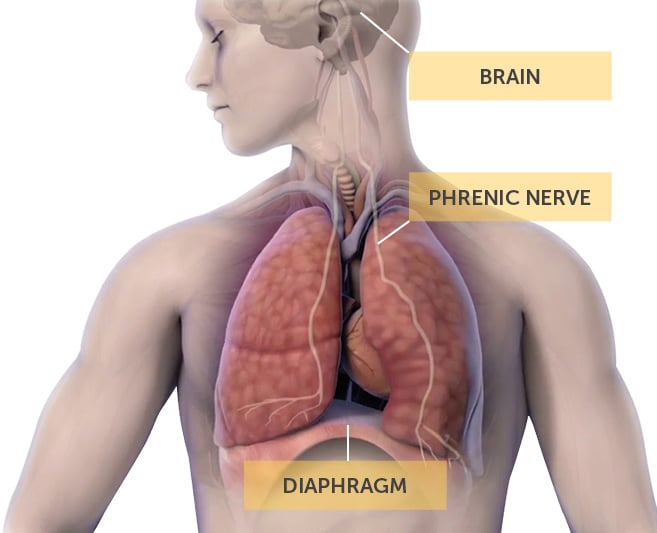
remedē uses a pacemaker-like device to send regular signals to the phrenic nerve that activate your diaphragm. The signals contract your diaphragm, pulling air into your lungs.1 remedē activates automatically each night based on programming to meet your specific needs. It turns on when:1
- It is within your desired sleeping hours (e.g. 11PM – 6AM), AND
- You are in a reclined position, AND
- You are not moving
If you roll over, sit up, or get out of bed, the therapy will pause and resume once the above three
conditions are met.1
Some examples can be helpful to understand when remedē activates:
Example 1: Jesse’s sleep hours are programmed to begin at 10pm, but he stays up talking
with friends until 11pm. remedē won’t activate until Jesse settles into bed at 11pm. Even
though the therapy is scheduled for activation at 10pm, it will sense that Jesse is up and
moving, so it will not turn on until he is lying in bed.
Example 2: Jesse is in bed reading at 11pm. It depends on Jesse’s position. If Jesse is fully reclined while reading, he may feel remedē begin to activate. If he is sitting up, he will not. If he does feel the therapy begin to activate, he can sit up a little higher to pause the therapy until he is ready to go to sleep.
Example 3: Jesse gets up at night to use the bathroom. remedē will pause when it senses that Jesse has gotten up or started moving. Once he settles back into bed, therapy will wait about 15 minutes before beginning to ramp up slowly. This will give him time to get back to sleep.
FDA status and clinical evidence
Pivotal trail and FDA approval
Phrenic nerve stimulation was FDA-approved on October 6, 2017 for moderate to severe central sleep apnea in adult patients.1
This approval was granted following a prospective, multicenter, randomized clinical trial that showed improvement in:
- SLEEP QUALITY
- OXYGENATION
- QUALITY OF LIFE
- APNEA-HYPOPNEA INDEX (AHI) AND
- CENTRAL APNEA INDEX (CA)
95% OF THE PATIENTS in the pivotal trial indicated they would have the system implanted again
Long Term Evidence
Additional publications have shown follow-up data at 1, 2, 3 years after implant. The benefits of the therapy remain consistent over this time period.5
Getting evaluated for remedē
Finding a Doctor
Only a doctor can help you determine if you are a candidate for remedē. It is recommended that you speak to a doctor who has experience treating patients with this therapy. A list of doctors offering remedē can be found here.
Diagnosing Your Central Sleep Apnea
Potential patients are evaluated for central sleep apnea during an overnight sleep study. The physician can then decide whether the patient is a candidate for phrenic nerve stimulation. If so, the physician and patient can discuss the proper course of action including the risks and benefits of phrenic nerve stimulation.
The remedē Procedure
The procedure to implant the device is typically performed on an outpatient basis by a cardiologist experienced in pacemaker placement.
It involves the implantation of the pulse generator in the upper chest and the placement of two wires into veins similar to a cardiac pacemaker. One lead delivers therapy via neurostimulation (stimulation lead) and the other passively senses breathing (sensing lead). The stimulation lead is located in close proximity to the phrenic nerve for stimulation of the diaphragm during treatment of CSA and the sensing lead provides feedback on demonstrated respiration when the patient breathes. While the implant procedure is generally considered safe, there are some risks that should be considered. As with any surgically implanted devices, these risks include, but are not limited to, pain, swelling, and infection.1
Following implant and recovery, the physician programs the pulse generator to the patient specific therapy needs.
Summary
remedē is an FDA-approved, implantable, non-mask therapy that effectively treats moderate to severe central sleep apnea in adult patients. It is designed specifically with central sleep apnea in mind, activating your breathing muscles to restore a normal respiratory pattern and rhythm. The first step in determining if you are a candidate for remedē is to speak to a doctor. Doctors offering remedē can be found here:




